|
1/6/2024 0 Comments Atom pictureWhy wouldn’t you want to take off the old glasses, put on the new ones, and use them all the time?” And now we actually have a really good pair. “Until now, we’ve all been wearing really bad glasses. “We want to apply this to everything we do,” Muller says. As a result, any lowered temperature that the researchers could reach may not reduce the movement of the atoms by a noticeable amount. 
It is theorized that atoms stop moving at absolute zero, but it has yet been impossible to reach that temperature. It is possible that the researchers could again beat their own world record by using material that is made up of heavier atoms, as they wobble less, or by cooling down the sample which also reduces the movement of atoms. “When we talk about temperature, what we’re actually measuring is the average speed of how much the atoms are jiggling.” “With these new algorithms, we’re now able to correct for all the blurring of our microscope to the point that the largest blurring factor we have left is the fact that the atoms themselves are wobbling, because that’s what happens to atoms at finite temperature,” Muller continues. By seeing how the pattern changes, the researchers were able to compute the shape of the object that caused the pattern. It generates images by processing multiple coherent interference patterns that have been scattered from a particular object and looks for changes in the overlapping regions. The microscope captures images using a computational method of microscopic imaging called Ptychography. It also solves a long-standing problem - undoing the multiple scattering of the beam in the sample, which Hans Bethe laid out in 1928 - that has blocked us from doing this in the past.” This opens up a whole lot of new measurement possibilities of things we’ve wanted to do for a very long time. We basically can now figure out where the atoms are in a very easy way. “It’s reached a regime which is effectively going to be an ultimate limit for resolution. “This doesn’t just set a new record,” Muller says. 
The results of this experiment have been published in a full report here. Immediately before 1913, an atom was thought of as consisting of a tiny positively charged heavy core, called a nucleus, surrounded by light, planetary negative electrons revolving in circular orbits of arbitrary radii.The resulting photo (above) depicts an electron ptychographic reconstruction of a praseodymium orthoscandate (PrScO3) crystal, zoomed in 100 million times. Direct experimental evidence for the existence of such discrete states was obtained (1914) by the German-born physicists James Franck and Gustav Hertz. Atoms absorb or emit radiation only when the electrons abruptly jump between allowed, or stationary, states. The Bohr model and all of its successors describe the properties of atomic electrons in terms of a set of allowed (possible) values. The Bohr model of the atom, a radical departure from earlier, classical descriptions, was the first that incorporated quantum theory and was the predecessor of wholly quantum-mechanical models. Understand how Neils Bohr refined the Rutherford atomic model in explaining the movement of electrons around the nucleus See all videos for this articleīohr model, description of the structure of atoms, especially that of hydrogen, proposed (1913) by the Danish physicist Niels Bohr. SpaceNext50 Britannica presents SpaceNext50, From the race to the Moon to space stewardship, we explore a wide range of subjects that feed our curiosity about space!. 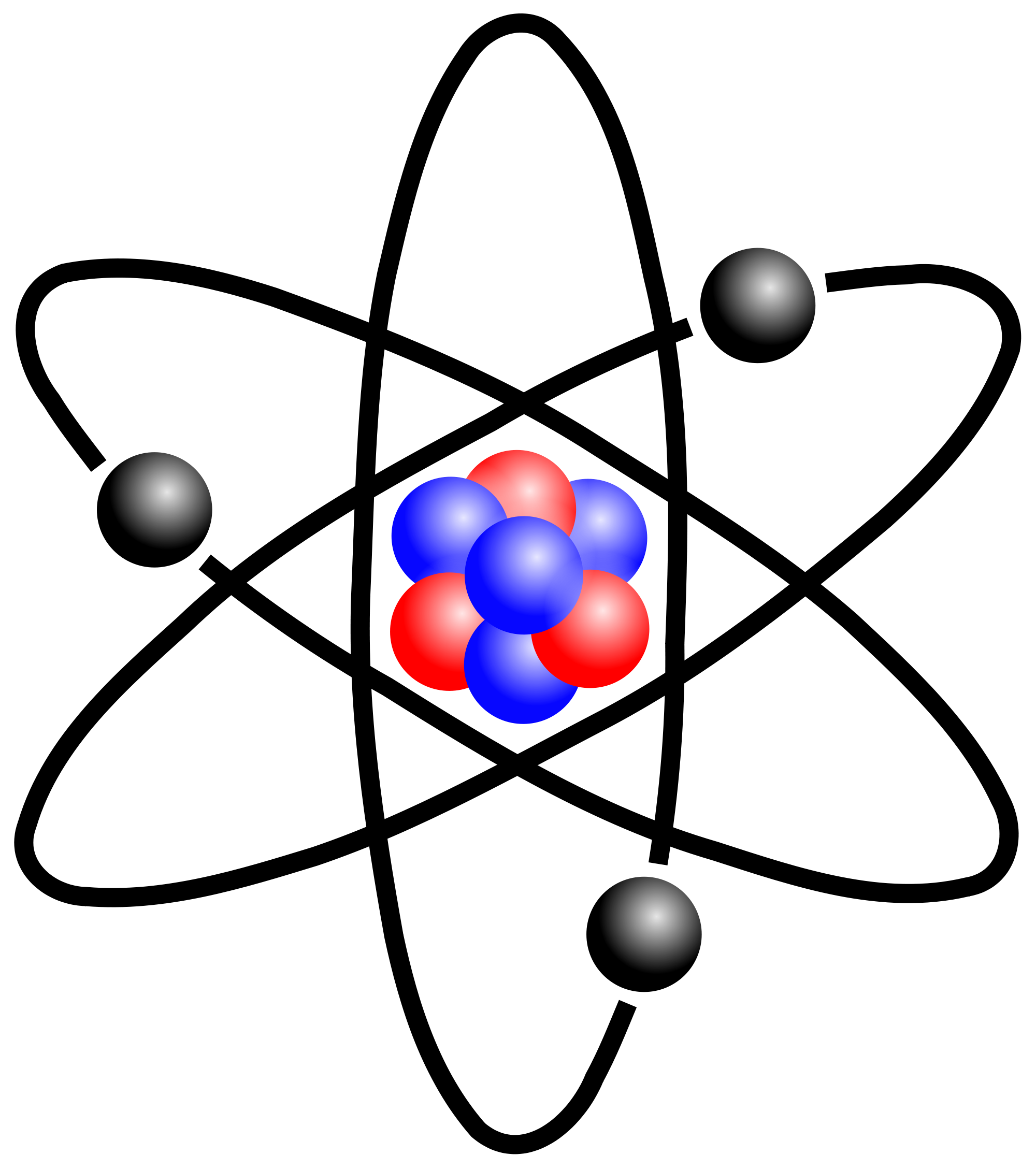
Learn about the major environmental problems facing our planet and what can be done about them!
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
